In a move that could help Indian companies ramp up production of Covid-19 vaccines currently developed/marketed by Moderna, Pfizer and Johnson & Johnson, Katherine Tai just released a statement supporting the waiver of Intellectual Property Rights (TRIPs provisions on the World Trade Organization), relating the production of these vaccines Tai,...
India’s Own Covid-19 Vaccine is 81 Percent Effective
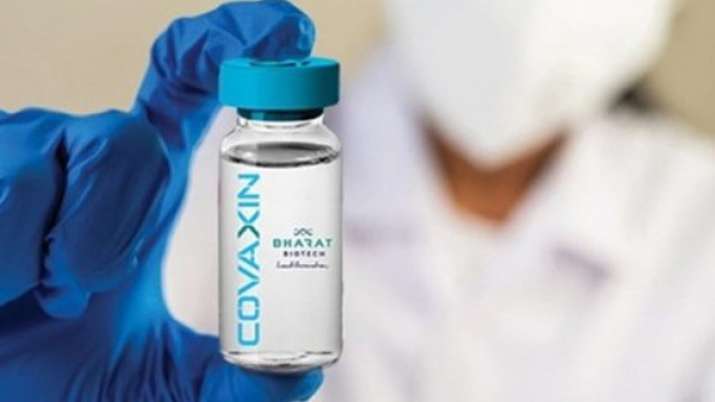
Yesterday, Hyderabad-based Bharat Biotech published interim results from the Phase 3 trial of it Covid-19 vaccine, branded as Covaxin. 25,8000 subjects participated in the trial. The trial found 43 cases of COVID-19 patients who showed symptoms ranging from mild to moderate and severe, and of the total cases, 36 were from...
Pfizer, India's Council of Medical Research to Address Antimicrobial Resistance
New York City-based Pfizer and state owned Indian Council of Medical Research (ICMR) have partnered to establish a center in New Delhi to combat antimicrobial resistance in India. The joint initiative will implement a series of comprehensive interventions, ranging from programs for training staff in the healthcare sector to scaling...
Biocon India Receives US FDA Approval for Biosimilar
India's Bangalore-based Biocon Ltd is the country's first company to have a "biosimilar" approved in the U.S. for Roche’s drug Trastuzumab, marketed as Herceptin, and used in treating certain breast and stomach cancers. Kiran Mazumdar-Shaw, the company’s chairperson and managing director said, "This was the first biosimilar for Herceptin approved...
Global Healthcare Companies Scout for India Startups
Earlier this year, The India Export reported that Netherlands-based Philips was in the process of creating a venture fund to invest in Indian health-tech startups. And indeed, companies such as Roche, Johnson and Johnson, Pfizer and Medtronic among others, are also scanning India for startups that align with their portfolios....